The only speech-based biomarker platform for clinical trials with advanced proprietary behavioural analytics for multiple disease areas and use cases with patient-centric intake protocols enabled by agentic AI.

Manual patient diaries and static rating scales collect data only at scheduled checkpoints, missing critical changes between visits.

Patients record 2-3 minutes of voice daily. Speech biomarkers detect cognitive, mood, and behavioural changes in real time.
Traditional PRO collection relies on patients completing written questionnaires between site visits. This creates inconsistent reporting due to recall bias, low compliance from burdensome formats, and delayed insights that arrive too late for adaptive trial decisions. Critical dropout signals and treatment effects remain invisible until the next scheduled checkpoint. Most methods also fail to catch objective observable and unconscious behaviours.

Depending on the protocol, patients record 2-3 minutes of natural speech daily through voice journaling or provide input via more thorough interviews or conversations with agentic AI. Traditional surveys can also be automatically facilitated through the speech interface.
AI analyses acoustic features (pitch, speech rate, pauses), semantic patterns (sentiment, coherence, vocabulary) and behavioural cues such as protocol adherence to extract behavioural and emotional biomarkers. This provides real-time visibility into behavioural, cognitive and mood changes as they happen.

Continuous monitoring identifies dropout risk before patients disengage, enabling proactive retention strategies. Earlier detection of treatment response allows for faster interim decisions and shorter trial durations. Objective, comparable data across all patients strengthens regulatory packages and accelerates approval timelines.
Agentic AI and guided journalling to extract natural speech data. GenAI-powered conversational AI enables natural discussions through voice or text. Curated guided journalling programmes support trial monitoring and provide feedback based on behavioural data.
Behavioural and emotional speech biomarkers for personalised insights. Analyses psychosocial expressions and behaviours, detects emotions using Plutchik's classification model, and extracts paralinguistic features including speech rate, length of phrases, interruptions, and pauses.
Proprietary AI algorithms for automating clinical psychometric assessments. Analyses patient sessions, interviews, and journal entries with custom LLMs for correspondence to psychometric scales. Provides psychometric progress analytics following individual behaviour over time.
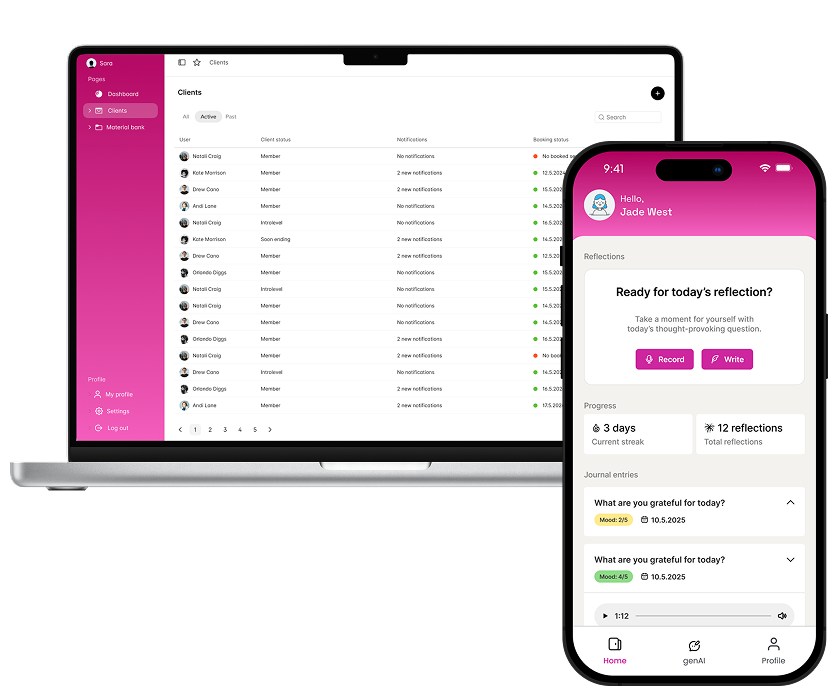
Low-friction voice journalling with guided prompts. Keeps patients engaged whilst collecting high-quality speech data in real time.
Real-time behavioural insights for clinical teams. Monitor trends, receive alerts, and track engagement across cohorts.
Create and validate custom speech-based endpoints. Design new behavioural biomarkers tailored to your study protocol or therapeutic area.
Integrate into existing trial infrastructures. Connect Adalyon's behavioural intelligence capabilities with your current systems and workflows.
%20(1).png)
The platform extracts key behavioural patterns from speech and immediately discards the original recording. All insights are non-personally identifiable data that cannot be traced back to individual voices.
The platform complies with GDPR and the EU AI Act. DPIA reviews and DPA documentation are available for audit.
Patients can easily manage their consent through built-in tools, ensuring ethical and regulatory compliance for therapy and clinical trials.
Information Security Management System (ISMS) aligned with ISO 27001 standards. The platform maintains enterprise-grade controls across access, availability, and data protection.
The platform continuously monitors and reviews systems, suppliers, and third-party tools in line with ISO 27001 control objectives.
Microsoft Azure hosts all data within the EU. Regional data residency, strict access controls, and advanced security measures protect all information.
Our team combines expertise in AI engineering, clinical research operations, and behavioural science. We've spent years developing speech analysis algorithms and behavioural analytics—we understand both the technical challenges of extracting reliable biomarkers and the regulatory requirements of bringing digital endpoints to market.

Ulrik previously worked as Director of Global Data and AI at Novo Nordisk, where he led initiatives across clinical use cases and trial design. With a background in finance, data analytics, and mathematics, he brings deep expertise in translating complex data into actionable clinical insights.

Katarina has over 20 years of experience spanning digital and IT services, healthcare, and HRM/wearables. Her academic background in behavioural science and information systems positions her at the intersection of technology and human behaviour, driving Adalyon's scientific approach to speech-based biomarkers.

Riko previously worked at Apple on Siri speech solutions and conducted behavioural analytics research at Stanford University. As a tech lead with emphasis on back-end architecture and AI/ML & NLP, he architects the technical foundation that powers Adalyon's speech analysis platform.

Herman is an enterprise-grade architect and senior team lead who masters the full solution stack at Adalyon. With previous experience in fintech and behavioural analytics, he ensures the platform meets the rigorous security, scalability, and reliability standards required for pharmaceutical clinical trials.


Heikki brings extensive experience from his roles as an Associate Partner at McKinsey focusing on Life Sciences & Healthcare, a digital health entrepreneur, and clinical work. He has vast experience in leading projects in clinical trial design, investments, and strategy for pharma and biotech clients.

Kane is the Director of Psychology at Egon Zehnder, a global leadership advisory and executive recruitment firm. He has a background in organisational psychology and enjoys creating new solutions combining AI and organisational psychology. Kane has extensive, international experience in organisational development, leadership assessments, and radar on the international HR tech market.
.png)
Contact us to discuss how speech biomarkers can fit your trial design. We'll walk through your therapeutic area, endpoints, and integration requirements—then show you exactly how the platform delivers continuous behavioral insights without adding patient burden.